Research Article - (2022) Volume 10, Issue 1
Mardan premier sugar plant is one of the biggest sugars industry in Pakistan. Because of non-adoptability of national environmental quality standards for the treatment of wastewater, a critical ecological and wellbeing concerns is available. A lot of poison loaded wastewater delivered at different phases of sugar creation. Like creating effluents during stick smashing and refinery activity; that is risky, harming and corrupting the ecological biological system. For the current examination, inspecting of water effluents was directed at and analyzed for pH, Total Suspended Solids (TSS), all Total Disintegrated Solids (TDS), Biochemical Oxygen Interest (BOD), Chemical Oxygen Demand (COD), alkalinity, chloride, electrical conductivity, smelling salts, copper, zinc, sulfate, phosphate, calcium, magnesium, shading and temperature. The logical information uncovered that the noticed estimations of TSS, BOD, COD, pH and smelling salts were higher than Pakistan’s National Environmental Quality Standards (NEQS). Those boundaries which are not in the scope of admissible cutoff points are TSS 249 mg/ L, BOD 4492 mg/L, COD 9022.67 mg/L, pH 5.3 and alkali 48.2 mg/L.
The investigation investigates the wellbeing and natural effects of waste water discharges from the sugar business, especially as far as contamination boundaries that are found to exceed NEQS; It likewise evaluates the specialized and administrative setting where the wastewater issue might be handled, talking about existing ecological approaches and enactment and accessible choices including innovations for the decrease of wastewater volume and contamination load, end of line treatment, and reusing/reuse of waste water.
Wastewater characteristics, NEQS, Water quality, Sugar mills
1.1. General/Background
The sugar industry in Pakistan is the 2nd largest agro based industry after textile. Pakistan is an important sugar cane producing country and is ranked 5th in world cane acreage and 9th in sugar production. The share of sugar industry in value added of agriculture and GDP are 3.2 percent and 0.7 percent.
This study represents the environmental aspects of the premier sugar mills and distillery company limited, Mardan. It came into existence in 1949. The premier sugar mill is located on Nowshera road in district Mardan. It was commissioned in 1950 as the largest sugar factory in south Asia and has added a sugar beet plant to complement the cane plant [1-5].
In mills the sugar cane crushing season starts in November, and continuous till March. The crushed capacity of the mill is about 4500 tons/day. An alcohol distillery was set up in 1958 and planning for a citric acid plant is underway.
The working staff comprises about 1,500 workers both skilled and unskilled. It has about 500 skilled workers and the rest are unskilled. When the mill is closed its cleaning of the machinery and overhauling are undertaken in the presence of skilled workers.
The mill is situated on a vast area in Mardan and it has also extraordinary machinery which makes it superior from all other sugar mills in the country. The mill produces sugar and alcohol from sugarcane and sugar beet. The mill also has a well established laboratory for checking the quality of sugar and to find different environmental parameters.
1.2. Sugar Cane Production in Pakistan
Sugar cane is cultivated from Khyber Pakhtunkhwa (KPK) to upper (Punjab) and lower (Sindh) plains of Indus is cultivated. In all the sugar cane producing regions, rainfall is low therefore canal irrigation system is important for sugar cane cultivation in Pakistan.
The main sugar cane growing regions are given below:
KPK: The main sugar cane growing areas in KPK include the districts of Char Sadda, Mardan and Peshawar.
Punjab: Punjab is the biggest and most important region of sugar cane cultivation and has 69 percent of total production of sugar cane in the country. The districts of Punjab important in this respect are Rahim Yar Khan, Bahawal Nagar, Bahawalpur, Layya, Muzaffar Nagar, Pak Pattan, Vehari, Sahiwal, Okara, Kusur, Mandi Bahauddin, Jhang, Toba Tek Sing, Faisalabad, Bhakkar and Sarghodha.
Sindh: The areas of Sindh where sugar cane cultivation is concentrated are the districts of Thatta, Badin, Hyderabad, Mirpur Khas, Sangarh, NaushehroFeroz, Nawab Shah and Khairpur [6-10].
1.3. Sugar Mills Capacity
In Pakistan 78 sugar mills (43 in Punjab, 29 in Sindh and 6 in KPK) are operating having crushing capacity of 361,300 tons of cane per day (TCD). Based on 160 days season these sugar mills have a total crushing capacity of 58 million tons of sugarcane capable to produce 5 million tons of refined sugar and 3 million tons of molasses.
In order to enable the country to be self-sufficient in sugar, a minimum area of 1.150 million hectares should remain under sugarcane cultivation, which can produce 50 million tons at an average yield of 43.86 tons per hectare. In Pakistan present sugarcane varieties are not yielding reasonable production and led to the prevailing sugar crisis. The government may consider importing high yielding varieties of sugarcane for averting any sugar crisis in the country for the future (Table 1).
| Countries | 1989-1990 | 1991-1992 | 1996-1997 |
|---|---|---|---|
| USA | 77.44 | 76.8 | 75.19 |
| Mauritius | 70.33 | 72.83 | 71.39 |
| India | 66.06 | 64.14 | 70.3 |
| Brazil | 62.02 | 61.74 | 67.42 |
| Pakistan | 41.55 | 43.38 | 43.54 |
| Cuba | 59.98 | 57.04 | 34.83 |
Table 1: Sugarcane yield per hectare per ton in various.
1.4. Objective of Study
The wastewater of the premier sugar mill Mardan discharges into an open drain. When it discharges into the river without treatment, it has a high strength of bod and cod which is very dangerous for the aquatic life as well as for human health. The study conducted for the following objective [11-15].
• To analyze characteristics of waste water of sugar mill Mardan.
• To find the strength of different wastes included in this water.
• To compare the results with the standards of NEQS.
1.5. Sugar Industry Background
General/Company background: Sugar is one of the most important substrates for human diet and it is an essential product of the human life. Sugarcane is a valuable crop for bio products because it produces sugar which has very high demand in the market and also bagasse which provides energy in the form of fuel for the generation of electricity and steam. Bagasse is used as input resource in 80 sugarcane producing countries. In the past, sugar industry produced only sugar but nowadays sugar industries are involved in the production of sugar, electricity and ethanol. So, sugar industry is now called as the cane industry.
The top five nations, viz., India, Brazil, Thailand, Australia and China accounted for nearly 40% of the total sugar production in the world. Sugar is produced approximately in 115 countries in the world. Out of these, 67 countries produce sugar from sugarcane, 39 from sugar beets and 9 countries from sugarcane as well as sugar beets. In other words, sugar is produced 70% from sugarcane and 30% from sugar beet and cassava, etc. In the world, Brazil and India are holding first and second rank in sugarcane production countries, respectively. Though, each produces 275 million tons of sugarcane.
India is the second largest producer of sugar in the world. In the year of 2012-2013, 526 mills are operated in India, which produced 33.90 million tons of sugar.
Sugar industry is basically seasonal in nature and operates only for 150-210 days in a year (November to May). A significantly large volume of waste is generated during the manufacture of sugar and contains a high amount of pollution load, particularly in terms of suspended solids, organic matter, and press mud, bagasse and air pollutants [16-20].
Several chemicals are used in sugar industries mainly for coagulation of impurities and refining of end products. Ca (OH)2 is used to clarify and to increase pH of juices. A small quantity of H3PO4 is added prior to liming to improve clarification. CO2 gas is bubbled through the defected juice to lower pH, which result in the improvement of precipitation of impurities. Polyelectrolytes, which are polymer based chemical, are also used for coagulation impurities during defection and carbonation process. SO2 is bubbled through the defected raw sugar to remove colour. Dilute solution of NaOH or Na2CO3 is used for the periodic descaling of heater followed by neutralizing it with dilute HCl. Lead sub acetate is used for the analysis of sugar content. These entire chemicals, one way or another, are contributing towards increasing the organic strength, dissolved solid and suspended matter.
Industrialization is considered the cornerstone of development strategies due to its significant contribution to the economic growth and human welfare. It has become a yardstick for placing countries in the league of nations and an index of its political stature. Industrialization, like other human activities that impact on the environment, often results in pollution and degradation. Industries turn out wastes which are peculiar in terms of type, volume and frequency depending on the type of industry and population that uses the product.
Environmentalists and scholars in other disciplines have attempted to examine the impact of industrial activities on the physico-chemical parameters and heavy metal concentration of rivers/streams, while others carried out a bacteriological assessment [21-24].
A scheme showing the water flows in a sugar process production is appeared in Figure 1. During the sugar production water flow in mill is appeared in Figure 1 and water leaving the mill in following ways: (a) Together with the products of the mill: in molasses and in filter cake. (b) In the form of vapor in: boiler gases, which may or may not be saturated, depending on whether a wet scrubber is used; vapor evaporated in cooling towers or spay pond; flash vapor from the heated juice flash tank; evaporation from diffuser or mills. (c) In liquid form as: surplus condensate; overflow from the cooling water circuit; boiler blow down; drift loss from the cooling towers or spray pond; effluent (wash down or spillage) from drains.
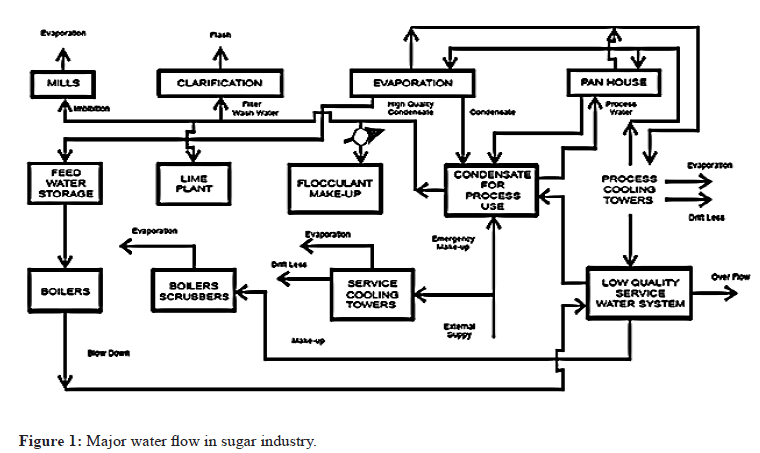
Figure 1: Major water flow in sugar industry.
1.6. Process Description
Manufacturing process of sugar: Sugar manufacturing process in the industry consists of the following units. Majority of units produce various waste water along with solid waste and air pollution.
• Weighing
• Cane carrier and cutter blades
• Mills or Mill house
• Boiling house
• First carbonation tanks
• Pressure house
• Second carbonation tanks
• Sulphonation chamber
• Evaporators
• Pans
• Separators
• Packing
Weighing: First of all, the sugar cane brought to mills by trucks and trolleys is weighted by electrical balance which gives directly the weight of loaded trucks and subtracting the weight of empty truck. In this way the sugar cane weight can be calculated.
Sugarcane carrier and cutter blades: The trucks are unloaded by labour in a ditch like structure which contains a belt at the bottom. The function of the belt is to take the sugar cane to the cutting machine. The belt is an automatic one which can carry heavy load of sugar cane this belt is called cane carrier. The belt ends at a tunnel where sharp blades cut the cane into pieces (Figure 2).

Figure 2: Sugarcane carrier and cutter blades
Mills or mill house: The sugar cane is crushed in the mill house, which results in trickling down of sugar can juices into a pool. The mills operate with the help of very big rotating wheels and so much pressure is applied on the sugar cane by huge rollers of the mills that all of its juice comes out. The mills are six in series and each has different types of teeth. Finally, the cane is emptied of juice and substance called bagasse is left. It is carried to the boiler house by the use of belt. Bagasse is its solid waste (Figure 3).

Figure 3: Mill house.
Boiling house: After crushing the sugar cane and extracting its juice, is then transferred to the boiling house. Here the juice contains much water therefore its necessary to evaporate the water from it. The juice is boiled in different stages. In first stage temperature ranges from 50℃-60℃ and in second stage its ranges from 80℃-85℃. After this process the juice is boiled and the steam comes in pipes (Figure 4).

Figure 4: Boiling house.
First carbonation tank: After evaporation of water, the juice comes to the first carbonation tank. Lime and carbon dioxide is added in this tank. In this stage the pH value of the sugar is maintained in the range of 10-10.5.
Pressure chamber: The filtration of the juice takes place in pressure house where temperature is kept same as that of first carbonation tanks. Mud separation takes place in the pressure house. After passing the juice into number of filters (cloths). At this stage water along with inorganic particles are mixed with producing effluents of high strength of wastewater.
Second carbonation tanks: After separation of the mud the clear sugar cane juice comes to the second carbonation tanks where further carbon dioxide is added to it to reduce the pH value from 10. Further filtration is done to remove the dirt, causing generation of wastewater. In this process the pH reduces to 8-8.5 (Figure 5).

Figure 5: Carbonation chamber.
Sulphonation chamber: After the filtration the juice comes to the sulphonation chamber where Sulphur dioxide is added to it. Sulphur dioxide is obtained from the burning of the Sulphur. The Sulphur is burnt in kiln whose chimney is directly attached to the sulphonation chamber. Sulphur is added for better shining (Figure 6).

Figure 6: Sulphonation chamber.
Evaporators: Evaporator takes place in evaporators where the temperature is 110℃. The viscosity of sugar juices is increased and the juice now called syrup. This juice is now transferred to the storage tanks (Figure 7).

Figure 7: Evaporation chamber.
Pans: The pans are three in numbers having dumb like shape. The syrup is further boiled in these pans and different sizes of sugar grains are formed (Figure 8). In first pan the sugar called A-sugar having smaller size and much sweeter is obtained while boiling is continuous. In Second pan further boiling and the reduction of sweetness occurs and the grain size increase. Sugar at this stage is called B sugar.
Figure 8: Pan chamber.
In third pan continuous boiling take place and the size of grains further increases and the sweetness reduces to normal. This sugar is in the form of crystals and not pure, called C sugar.
Separators: The crystals of sugar obtained from pan C are not used but it is dropped over crystallizer in which the crystals are broken down. Then the sugar is passed through separation tanks called centrifugal separation in which it is soaked and grade of round shape are obtained. It is again passed through centrifugal where it gains white colour then it is passed to sugar drier. The sugar is passed in layers and is dried with help of fans (Figure 9).

Figure 9: Separator.
Packing: Sugar after drying is dropped in sugar sheets having cone like shapes and is packed automatically (normally 50 kg bags) (Figure 10).

Figure 10: Packing.
1.7. Sugar Processing Chemicals
To obtain the purest and most refined form of the desired product from the raw materials, involves the use of a number of chemicals which eventually find their way into the wastewater stream. The following are the main chemicals used:
LUKOCIDE-BCS (Organic mill sanitation biocide registered patent product): Lukocide BCS is a highly concentrated powerful biocide, scientifically developed for highest sugar recovery by way to inhibit and stop the growth of bacteria and fungi or other biomass during the milling process in sugar mills. This biocide maintains purify of sugarcane juice especially from all strains of leuconostoc, Lactobacillus and other microorganism are effective at very low dosage. It is highly stable over wide range of pH and temperature, controls drop in purity and cost effective, self-degradable and eco-friendly.
LUKOCIDE-QUAT (Microbicide): It is highly effective quat base bactericide for mill sanitation specially designed for sugar industries. It inhibits and eradicates the growth of bacteria, fungi and other micro-organism, leuconostoc as are met during the milling process in sugar mills.
VISRAL-VRS (Synthetic viscosity reducer): VISRAL-VRS: It is a concentrated latest improved product to increase the fluidity of molasses stickiness, and excellent wetting out agent. VISRAL-VRS is a boiling aid specially formulated to reduce the viscosity of syrup massecuites and can be used throughout in whole season without corrosion of instruments.
LUKOCIDE-DTC (Dithiocarbamate base mill sanitation chemical): LUKOCIDE-DTC is a specially designed formulation for high performance dithiol-carbonates. It is highly effective against all types of microorganism found in cane juice, including seine producing bacteria and yeasts. LUKOCIDE-DTC effectively arrests the microbial sucrose inversion and controls the production of polymers in the cane juice and improves the sugar quality.
LUKOCIDE-OSC (Organo sulphur base mill sanitation biocide): LUKOCIDE OSC is a liquid organo sulphur base product specially designed for deducing sugar loss, due to microbial inversion. LUCK-OCIDE-OSC is odorless, non-toxic at usage concentration, non-corrosive to sugar mill equipment’s, stable over wide range pH and temperatures effective at very low dosage.
LUFLOC-810 (High molecular weight locculent): Anionic flocculent LUFLOC-810 is high molecular height polymer flocculent suitable for sugar cane juice classification and filtration mud slurry. It is waste soluble non-toxic polyelectrolytes with powerful flocculating properties.
Sampling
Sampling is one of the most important phases of water quality tests. Therefore, extreme precautionary measures are taken.
Sampling Types
There are three main types of sampling that are grab samples, composite samples and integrated samples. Each of them is described below:
Grab Samples
It refers to the samples taken at the selected locations, depth and time and then analyzed for the constituents of interest. It represents only the composition of the system at that time and location.
Composite Samples
It refers to the mixture of grab samples collected at given locations at different times.
Integrated Samples
Integrated samples may be throughout composite in depth and width rather than time. The grab samples collected from various points at different depths across the width may be mixed in proportion to the relative flows at those points.
Sampling Method
• Distilled bottles of 1.5 liter capacity were used for collection of Mardan sugar industrial sewage.
• Four different sites were selected for collection of samples of the sugar mills i.e. lime killer, steam boiler, mill house and a mixed sample where all the three sewages combined.
• The bottles were properly washed by sample of selected sewage two three times, and then completely filled expelling air completely and closing the bottles with a cork.
• The samples were then carefully brought to the laboratory for analysis (Table 2).
Laboratory Work
| Parameters | Method used | Instrument used |
|---|---|---|
| pH | ------- | pH meter |
| Chloride | Argentometric method | ------- |
| Ammonia | Titrimetric method | ------- |
| Conductivity | ------- | Conductivity meter |
| Alkalinity | Titrimetric method | ------- |
| BOD | Winkler modification | ------- |
| COD | Open reflux method | DR-2000 Spectrophotometer |
| TS | Gravimetric method | ------- |
| TSS | Gravimetric method | ------- |
| TDS | Gravimetric method | ------- |
| Temperature | ------- | Thermometer |
| CU | ------- | DR-2000 Spectrophotometer |
| Zn | ------- | DR-2000 Spectrophotometer |
| Sulphate | ------- | DR-2000 Spectrophotometer |
| Phosphate | ------- | DR-2000 Spectrophotometer |
| Temperature | ------- | Thermometer |
| Ca | ------- | DR-2000 Spectrophotometer |
| Mg | ------- | DR-2000 Spectrophotometer |
| Color | ------- | Nesselerizer |
Table 2: Methods and instruments used for waste water analysis.
2.1. Test for pH
Apparatus: Digital pH meter.
Procedure
• Take sewage sample in a beaker.
• Standardize the instrument with buffer solution (pH 4.2 and 7).
• Dip the electrode in distilled water after cleaning with tissue paper again dip it into sample and read the value of pH from display of pH meter.
Caution: The dipping of electrode in distilled water before and after dipping in sample is necessary for accuracy.
2.2. Test for Chloride
Apparatus: Burette with burette stand and porcelain title, pipettes with elongated tips, pipette bulb, conical flask, titration flask, 250 ml graduated cylinder, standard flask, wash bottle, beaker.
Chemicals: Potassium Chromate (K2CrO4) (indicator) and standard Silver Nitrate (AgNO3).
Procedure
• Fill the burette with silver nitrate solution of 0.0141 N. Adjust to zero and fix the burette in stand.
• Take 50 ml of the sample in clean 250 ml conical flask.
• Add 0.5 ml potassium chromate indicator to get yellowish color.
• Titrate the sample against the silver nitrate solution until the color changes from yellowish to reddish brown colour, i.e, end point.
• Note the volume of silver nitrate added.
• Repeat the procedure for other samples.
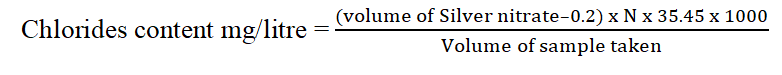
2.3. Test for Ammonia
Apparatus: Spectrophotometer.
Chemicals: Rochelle salts, nessler reagent.
Procedure
• First turned O N the spectrophotometer and enter the stored program number for ammonia nitrogen i.e. 380 and pressed read/enter button.
• Display will show: Dial nm to 425. Rotate the wavelength dial until the small display shows: 425 nm. And pressed read/enter button.
• Fill a 25 mL graduated mixing cylinder to the 25 mL mark with sample (the prepared samle).
• Fill another 25 mL mixing graduated cylinder with deionized water (the blank).
• Add three drops of Mineral Stabilizer to each cylinder. Invert several times to mix. Add three drops of Polyvinyl Alcohol dispersing agent to each cylinder (hold the dropping bottle exactly vertical). Invert several times to mix. Pipet 1.0 mL of Nessler Reagent into each cylinder. Stopper. Invert several times to mix.
• Now pressed SHIFT TIMER. A 1 min reaction period will begin.
• Pour each solution into a sample cell.
• When the timer beeps, the display show: mg/l N NH3 Ness. Place the blank into cell holder. Close the light shield and pressed zero.
• Then placed the prepared sample into cell holder. And close the light shield and pressed Read/Enter button. The display will show the result in mg/L.
2.4. Test for Conductivity
Apparatus: Conductivity meter (having probed), water sample, beaker.
Procedure:
• Take a water sample in beaker.
• Switch on the machine.
• Now wash the probe with distilled water and dry it.
• Now press the conductivity button and set machine to the 2-20 millisimen/cm.
• Now press the 20 buttons (10% of 20 is 2) if the value is less than 2. Then press the 2 buttons (10% of 2 is 0.2) if the value is higher than 0.2 then it is the conductivity of the sample. If the value is less than 0.2. Then press 200 microsimen/cm.
• Repeat the experiment for different sample.
2.5. Test for Alkalinity
Apparatus: Stand, burette, funnel, conical flask, beaker etc.
Chemicals: Phenolphthalein indicator solution, bromo cresol red indicator, standard solution (H2SO4) having normality 0.02.
Procedure:
• Take 50 ml of water sample in a flask. Add six drops of phenolphthalein indicator in the sample (water), note the initial reading of the burette containing H2SO4 (N=0.02).
• Start the titration till the colour changes and note the reading of the burette, calculate the phenolphthalein alkalinity using the formula alkalinity=(final reading initial reading) × 100/50.
• Now add six drops of bromo cresol green methyl red solution which turns the colour to greenish one. Note the initial reading of the burette and start the titration till the colour changes to purple and note the final reading.
• Calculate total alkalinity by using the formula:
• Total alkalinity=(final reading-initial reading) N x 50 x 1000/wt. of sample.
2.6. Test for BOD
Apparatus: BOD incubator, burette and burette stand, 300 ml BOD bottles, conical flask, pipette, graduated cylinder
Reagents: Magnesium sulphate, alkaline iodide-azide solution, sulfuric acid (concentrated), sodium thiosulphate, starch indicator.
Procedure:
• Took six 300 ml glass stoppered BOD bottles (three for sample three for blank).
• Add 3 ml, 6 ml and 9 ml of sample to the three BOD bottles and fill the remaining quantity with the distill water.
• After the addition immediately place the glass stopper over the BOD bottles and note down the number of bottle for identification.
• Took three bottles from the six bottles and fined its DO at zero day and put the remaining bottles in a BOD incubator at 20℃ for five days.
• Add 2 ml of manganese sulphate in each 3 BOD bottles.
• Add 2 ml of alkali-iodide-azide reagent in the same manner precipitates will be formed.
• Add 2 ml of conc. sulphuric acid in the same way in each bottle.
• Carefully invert the sample several times to dissolve the floc.
• Then we removed 100 ml solution from each bottles and 1 ml starch indicator to it.
• Titration needs to be started immediately after the contents of the conical flask.
• Fill the burette with sodium thiosulphate, and fix the burette in the stand.
• Titrate the solution with standard sodium thiosulphate solution until the blue colour disappear to colourless.
• Note down the final reading and find the Dissolved Oxygen (DO).
• After five days take out the bottles from incubator and analyze the sample and blank for BOD.
• Also find the DO of the sample after incubation.
So BOD of the sample: BOD (mg/L)=((DO at zero day-DO at day five) × 300)/Volume of sample.
2.7. Test for COD
Apparatus: Spectrophotometer (0 mg/L-1500 mg/L), reflex apparatus, vails, 500 ml conical flask, pipettes with elongated tips, pipette bulb, wash bottles, a fluxing flask, sewage sample.
Chemicals: Standard K2Cr2O7solution (0.025 N), sulphuric acid (concentrated), standard ferrous ammonium sulphate solution (0.1 N), ferrous indicator solution, mercuric sulphate.
Procedure:
• Took three vials tube from box of 1500 mg/l which contain chemically potassium dichromate (K2Cr2O7), Sulphuric acid and Mercuric sulphate.
• Added 2 ml of waste sample in each of three COD vials and shacked them.
• Kept the vials in COD reactor for digestion and preheated to 150℃ for 2 hours.
• After completion of digestion removed the COD vials from COD reactor and cooled them.
• Turned ON the spectrophotometer and selected program number 435 and adjusted wavelength 620 nm.
• Placed COD vials adaptor in the cell holder and insert blank vial and pressed zero button. Put the sample vial in the cell holder and pressed “read enter” button.
2.8. Test for Total Solids
Apparatus: China dish, measuring cylinder, analytical Balance, oven, waste water sample.
Procedure:
• Weigh the china dish.
• Took 50 ml of waste water sample.
• Poured the sample into china dish.
• Place the china dish in an oven maintained at 103℃ for 24 hours.
• After evaporation, took the china dish and put it in desiccator for some time.
• Now weigh the china dish again and note reading.
• Subtract the weight of china dish from the weight of china dish+sample.
Calculator Total Solids (TS) as follows:
Total solids, TS (mg/l)=mg of solids in the china dish x 1000/(volume of sample).
2.9. Test for Total Suspended Solids
Apparatus: Filter paper, measuring cylinder, analytical balance, and oven, waste water sample, distilled water, tripod stand, conical flask, beaker, stirrer, and funnel.
Procedure:
• Weigh the china dish.
• Took 50 ml of waste water sample.
• Fixed the filter paper into the funnel by spraying water on filter paper.
• Filtrated the sample.
• After filtration, took the filter paper and placed it an oven at 103℃ for 30 minutes.
• Took the filter paper and put it in desiccator for some time.
• Now weigh the filter paper again and note reading.
Suspended solids=((weight of filter paper+sample)-weight of filter paper) × 1000/volume of sample
2.10. Test for Total Dissolved Solids
Apparatus: Filter paper, measuring cylinder, analytical balance, and oven, waste water sample, distilled water, tripod stand, conical flask, beaker, stirrer, and funnel (Table 3 and Figure 11).
Procedure:
• Weigh the china dish.
• Took 50 ml of waste water sample.
• Fixed the filter paper into the funnel by spraying water on filter paper
• Filtrated the sample.
• After filtration, took the filtrate in a china dish and placed it an oven at 103℃ for 24 hours.
• After evaporation, took the china dish and put it in desiccator for some time.
• Now weigh the china dish again and note reading.
• Subtract the weight of china dish from the weight of china dish+sample.
Calculator Total Dissolved Solids (TDS) as follows:
Total dissolved solids, TDS (mg/l)=mg of solids in the china dish x 1000/(volume of sample).
2.11. Test Results
Collection points
• Upstream sample.
• Effluents of sugar mills (main drain under G.T road).
• 5 meters downstream from confluence point.
• 15 meters downstream from confluence point.
| No | Parameters | Collection points (locations) | Average value | |||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |||
| 1 | pH | 4.6 | 5.6 | 5.8 | 5.7 | 5.4 |
| 2 | Chloride | 298 | 304 | 308 | 305 | 303.8 |
| 3 | Ammonia | 45.6 | 48.2 | 50.8 | 51.2 | 49 |
| 4 | EC | 1.63 | 1.81 | 1.867 | 1.85 | 1.8 |
| 5 | Alkalinity | 301.5 | 308.4 | 310.9 | 310.5 | 307.8 |
| 6 | BOD | 5120 | 4260 | 4096 | 4120 | 4399 |
| 7 | COD | 9002 | 9454 | 8612 | 8715 | 8945.8 |
| 8 | TS | 4196 | 4113 | 4083.4 | 4090.3 | 4120.7 |
| 9 | TSS | 196 | 223 | 328 | 305 | 263 |
| 10 | TDS | 4000 | 3890 | 3755.4 | 3775.5 | 3855.2 |
| 11 | CU | 1.55 | 1.5 | 1.3 | 1.45 | 1.5 |
| 12 | Zn | 0.15 | 0.135 | 0.18 | 0.175 | 0.2 |
| 13 | Sulphate | 419 | 418.5 | 415 | 413.5 | 416.5 |
| 14 | Phosphate | 1.89 | 1.82 | 1.81 | 1.76 | 1.8 |
| 15 | Temperate | 22.1 | 22.5 | 31.6 | 33.5 | 27.4 |
| 16 | Ca | 361 | 365 | 358.9 | 355 | 360 |
| 17 | Mg | 268 | 265 | 263.5 | 260 | 264.1 |
| 18 | Color | Dark yellow | Dark yellow | Dark yellow | Dark yellow | Dark yellow |
Table 3: Physicochemical parameters of sugar mills (industry) Mardan waste water.
pH
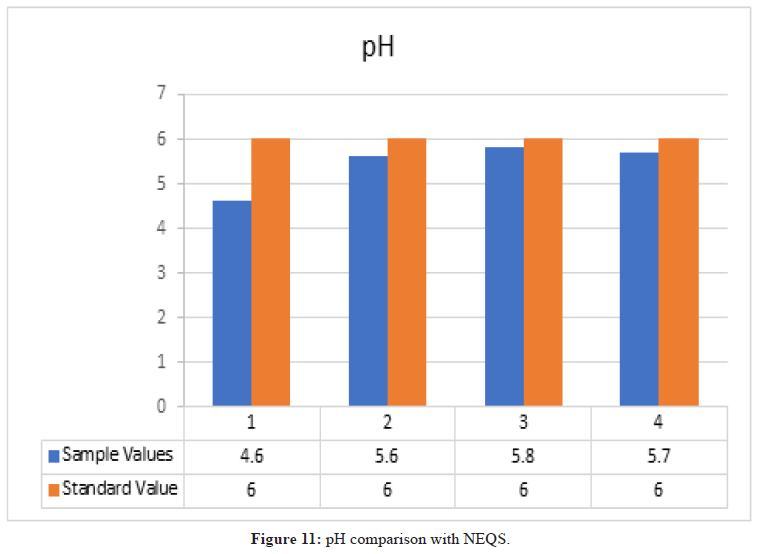
Figure 11: pH comparison with NEQS.
According to NEQS the value of pH should be 6-10 but the value from the test is 5.3, which is less than the permissible value so the effluent should be treated before disposal (Figure 12).
Chlorides
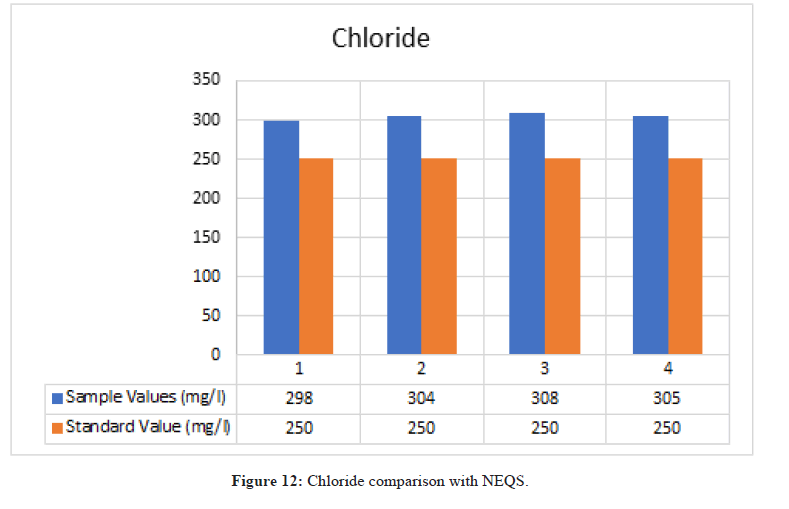
Figure 12: Chloride comparison with NEQS.
According to NEQS the value of chloride should be 250 mg/l but the value from the test is 303.33 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 13).
Ammonia
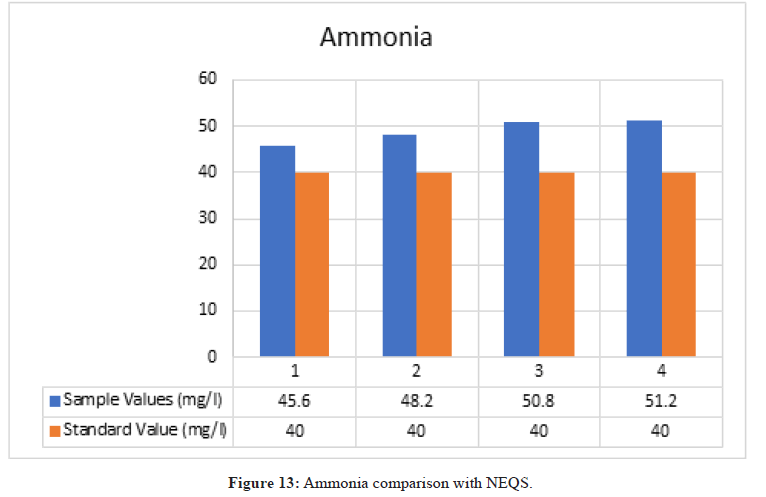
Figure 13: Ammonia comparison with NEQS.
According to NEQS the value of ammonia should be 40 mg/l but the value from the test is 48.2 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 14).
Conductivity Test
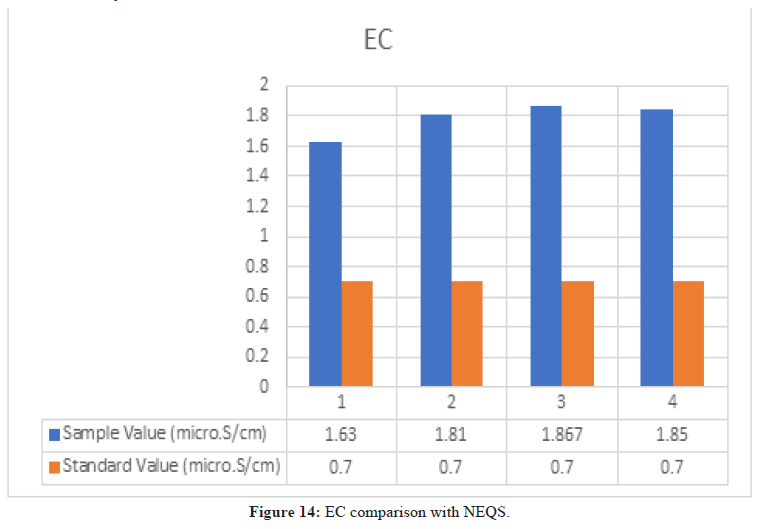
Figure 14: EC comparison with NEQS.
According to NEQS the value of EC should be 0.7 μS/cm, but the value from the test is 1.77 μS/ cm, which is greater than the permissible value so the effluent should be treated before disposal (Figure 15).
Alkalinity
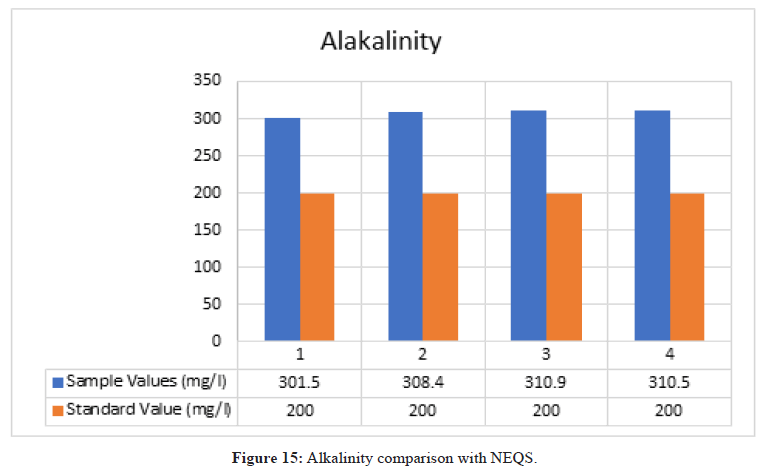
Figure 15: Alkalinity comparison with NEQS
According to NEQS the value of alkalinity should be 200 mg/l but the value from the test is 306.93 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 16).
Biological Oxygen Demand
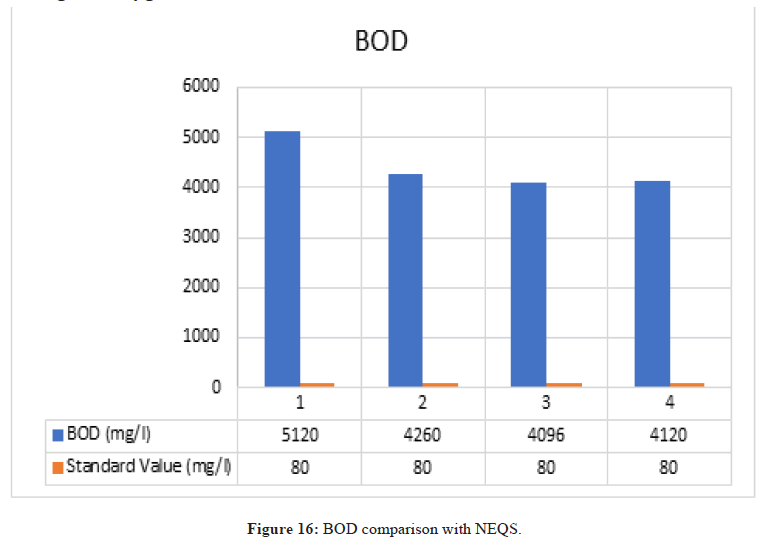
Figure 16: BOD comparison with NEQS.
According to NEQS the value of BOD should be 80 mg/l but the value from the test is 4492 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 17).
Chemical Oxygen Demand
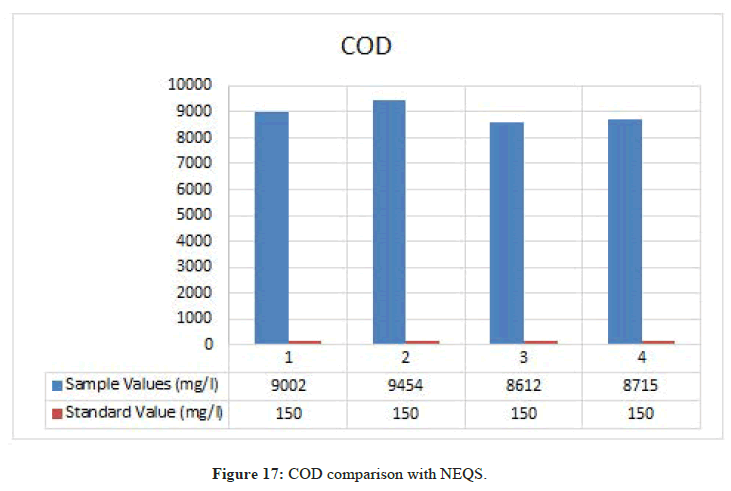
Figure 17: COD comparison with NEQS.
According to NEQS the value of COD should be 150 mg/l but the value from the test is 9022.67 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 18).
Total Suspended Solid
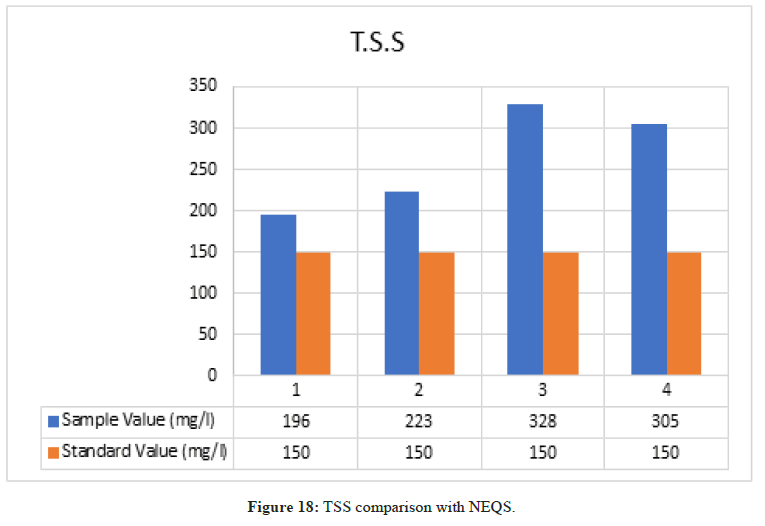
Figure 18: TSS comparison with NEQS.
According to NEQS the value of TSS should be 150 mg/l but the value from the test is 249 mg/l, which is greater than the permissible value so the effluent should be treated before disposal (Figure 19).
Total Dissolved Solids
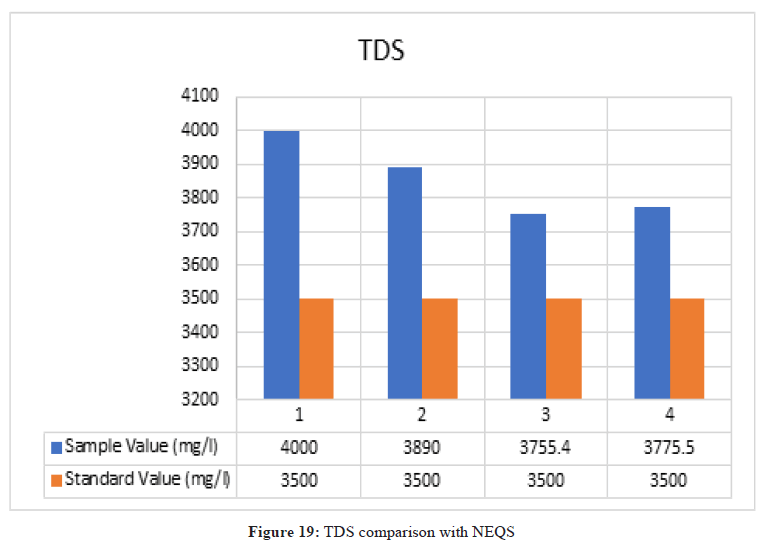
Figure 19: TDS comparison with NEQS
According to NEQS the value of TDS should be 3500 mg/l but the value from the test is 3881.8 mg/l, which is greater than the permissible value so the effluent should be treated before disposal.
• The waste water of the premier sugar mill has high strength of BOD and COD.
• This water is discharging through open drains, causing unhygienic conditions.
• This waste water is dangerous for aquatic life as well as for human health.
• This water can only be used for agricultural purposes.
• The water is discharging to the river without any treatment.
• People should be educated to mend their attitude towards the river.
• The sewer of the waste water should be discharged into the river after proper treatment.
• The solids waste should be disposed in a proper way.
• Laws should be enforced to uplift the situation.
Received Date: Dec 24, 2020 / Manuscript No: OAJOST-20-23755 / Editor Assigned: Dec 26, 2020 PreQC No: OAJOST-20-23755(PQ) / Reviewed Date: Jan 11, 2021 / QC No: OAJOST-20-23755 / Revised Date: Sep 23, 2022 Revised Manuscript No: OAJOST-20-23755(R) / Published Date: Sep 30, 2022 Doi: 10.11131/OAJOST.2022.10.002
Copyright: © 2022 S. Mulk. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.